If you have ever tried to fit regulatory text onto a tiny pouch label, you already know why ISO 15223-1 medical device symbols matter. Symbols save space, reduce translation headaches, and keep users from missing the one warning that actually keeps a patient safe.
I also see teams treat symbols like clip art, and that is where trouble starts. A sterile symbol used on a nonsterile device, or a do not reuse symbol missing from a single use product, is the kind of mistake that survives reviews and shows up on the production line.
ISO 15223-1 gives you a shared visual language for labeling, but you still have to apply it with discipline. The goal is not to pack every possible icon onto the box, it is to communicate the right constraints clearly and consistently.
This article focuses on practical use, not abstract theory, because labels get printed under deadlines. If you build a repeatable approach, you can keep compliance, readability, and brand consistency from fighting each other.
Why standard symbols are used on device labels and packaging
Medical device packaging has to communicate safety, traceability, and handling in a few square inches. Standard symbols let you say “single use” or “keep dry” without writing a paragraph in six languages.
ISO 15223-1 medical device symbols also reduce interpretation risk when products ship across borders. A consistent manufacturer symbol, for example, helps a hospital identify who made the device even when the brand name is unfamiliar.
Symbols are also a practical response to packaging constraints like small cartons, peel pouches, and blister packs. When you are fighting for space with UDI, lot, and expiry information, icons often keep the label from becoming unreadable.
There is a business angle too, because artwork changes cost money and time. If your team uses the same approved symbol set across product families, you cut down on redesign cycles and reduce errors when suppliers copy files.
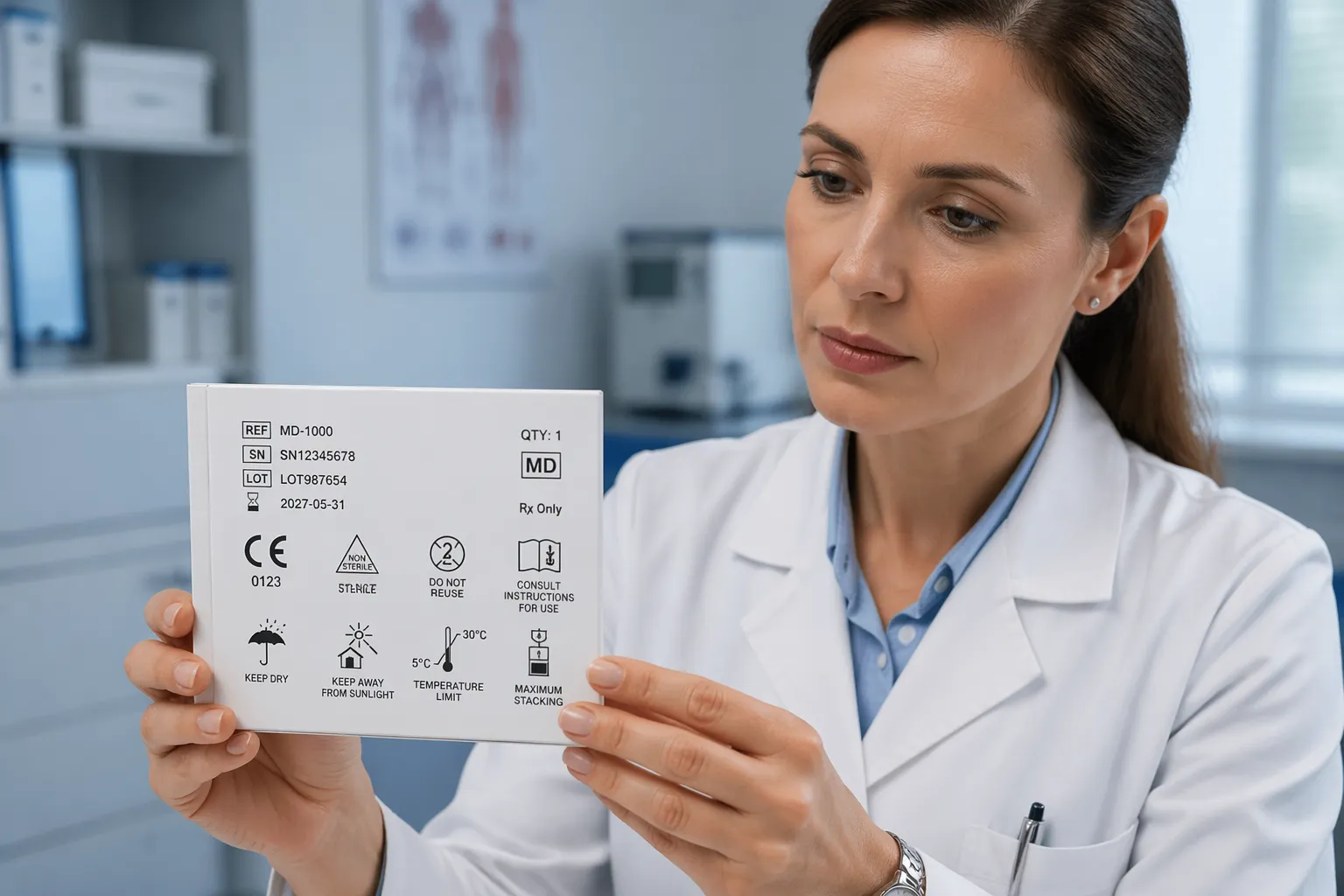
Standard symbols help when the product is handled by multiple people who never read the IFU. Receiving teams, sterile processing, and clinicians can all pull different decisions from the same small panel.
They also reduce the temptation to write micro text that becomes illegible after printing. If your only way to fit content is to shrink font size, you are already on the edge of a usability problem.
Symbols support faster scanning during time pressure, which is when mistakes happen. A user can spot a do not reuse symbol in a second, while a sentence about single use can get skipped.
They also make training easier because staff can learn a consistent set once and apply it across brands. In practice, many hospitals rely on symbol recognition more than manufacturer specific wording.
Another reason is change control, because symbols can be more stable than text across product updates. If your storage condition stays the same, you can keep the same icon even when marketing copy changes.
Symbols can also reduce the risk of mistranslation when you do need accompanying text. A translator may interpret a phrase differently, but a standard icon anchors the meaning for the user.
They are not a free pass, though, because the symbol has to be the right one and used in the right context. A symbol that is technically standard but applied to the wrong packaging level can still mislead.
When teams standardize symbols, they also standardize conversations across functions. Quality, regulatory, and packaging engineering can point to the same reference instead of debating personal preference.
Common ISO 15223-1 symbols and what they mean
Most label sets repeat the same core messages: who made it, whether it is sterile, whether it is single use, and how to store it. The sterile symbol is one of the most sensitive, because it ties directly to infection control and user trust.
The do not reuse symbol is another one that people underestimate, especially on devices that look durable. If a product is intended for single use, the symbol needs to match the IFU language and the risk file, not just the marketing story.
The manufacturer symbol looks simple, but it triggers real downstream behavior such as complaint routing and recall communication. When the legal manufacturer differs from a contract packer or distributor, the symbol placement and the adjacent address details need extra attention.
Other frequent symbols include batch code, use by date, date of manufacture, temperature limits, and keep away from sunlight. If you use ISO 15223-1 medical device symbols as a system, users learn your labels quickly and stop hunting for key data.
The batch code symbol matters more than people think because it is the key to traceability when something goes wrong. If your ERP prints the lot in a specific format, the symbol should sit close enough that users do not confuse it with another number.
The use by date symbol is often the first thing clinicians look for on sterile items. If the date is hard to find or ambiguous, the symbol cannot save you from a usability failure.
Date of manufacture is useful when shelf life is not the only concern, like when a product has a service interval or a warranty period. It can also help with complaint investigations when you are trying to correlate issues to a production window.
Temperature limit symbols are common on materials that degrade with heat or freeze damage. The mistake I see is listing a temperature range that is not supported by validation or shipping studies.
Keep dry is a simple icon, but it can be critical for devices with moisture sensitive packaging. If your sterile barrier can be compromised by humidity, that symbol is not optional decoration.
Keep away from sunlight shows up on products with light sensitive polymers or drug coated components. If the product sits in a clinic window for a week, that one symbol can prevent a performance complaint.
Do not use if package is damaged is one of those symbols that looks obvious until you watch someone open a dented carton anyway. If you include it, make sure the packaging design makes it easy to see damage in the first place.
Consult instructions for use is often used to keep the label clean while still directing users to critical details. The risk is that teams rely on it too heavily and push essential safety information out of sight.
Catalog number and serial number symbols help with ordering, servicing, and complaint trending. If you have both, keep them visually distinct so staff do not record the wrong identifier.
UDI related elements are not always ISO 15223-1 symbols, but they live in the same visual neighborhood. If you place UDI next to other codes, you need to prevent scanning errors and human transcription mistakes.
The bigger point is that each symbol has an operational consequence. If you cannot explain what action the symbol is meant to trigger, it probably does not belong on the label.
How to select symbols based on your packaging and use case
Symbol selection should start with the real world scenario, not a checklist copied from an older label. A sterile barrier pouch for an OR needs different emphasis than a nonsterile accessory in a clinic kit, even if both come from the same product line.
I prefer to map each symbol to a specific user decision, like “Can I reuse this?” or “Is this still within shelf life?” That approach keeps ISO 15223-1 medical device symbols from turning into a dense strip of icons that nobody reads.
Start by listing the packaging levels, because inner and outer packs do not serve the same user. The outer carton might need storage limits and traceability, while the sterile pouch needs sterility and barrier integrity cues.
Then look at how the product is actually stored and moved. A device that sits in a hot warehouse needs different emphasis than one that goes straight from production to a controlled hospital stockroom.
Consider whether the package will ever be separated from the IFU. If the pouch is used in a procedure room and the IFU stays in a binder, the pouch has to carry more of the safety load.
Think about the difference between “nice to know” and “must know before use.” If a symbol does not prevent a plausible misuse or support traceability, it is usually a candidate for removal.
Also decide which symbols belong on the shipper versus the sales unit. Shipping handling marks can be important, but mixing them with clinical symbols can create visual clutter and confusion.
For kits, be careful about applying symbols from one component to the whole kit. A kit can include a single use sterile component and a reusable nonsterile component, and the outer label has to avoid implying the wrong status.
For software or digital health accessories, the packaging might be minimal, but the symbol decisions still matter. If the physical label is small, you may need to push some information to an electronic label while keeping critical constraints on the device.
If you have multiple SKUs with the same packaging dieline, build a symbol baseline and then allow controlled variation. That keeps the label family coherent while still letting you reflect real differences like sterility or shelf life.
Finally, validate selection choices with people outside regulatory. If a warehouse lead or a scrub tech cannot interpret the label quickly, you probably selected the right symbols but arranged them in the wrong way.
| Packaging or context | Symbols that usually apply | Selection notes |
|---|---|---|
| Peel pouch for sterile device | Sterile symbol, use by date, lot, do not reuse symbol | Match sterilization method and barrier claims in the IFU |
| Blister pack inside a carton | Manufacturer symbol, lot, UDI carrier, temperature limits | Split symbols between inner and outer if space is tight |
| Reusable instrument tray | Reprocessable instructions reference, manufacturer symbol, catalog number | Avoid do not reuse symbol unless the device is truly single use |
| Home use device shipped direct to patient | Keep dry, fragile if applicable, temperature limits, manufacturer symbol | Prioritize handling and storage icons that prevent damage in transit |
Use the table as a starting point, not as a rulebook, because devices behave differently in the field. If your sterile pouch is always placed inside a protective carton, you may be able to shift some symbols outward without increasing risk.
When space is tight, decide which panel is the “decision panel” and protect it. Users should not have to rotate a pouch three times to confirm it is sterile and in date.
Be careful with duplicate symbols across levels, because repetition can either help or confuse. If the outer carton says sterile but the inner pouch does not, users may assume the pouch is generic and miss the sterility claim.
If you are using overlabels for regional needs, confirm the symbol set remains intact after overlabeling. I have seen overlabels cover the lot symbol or partially obscure the do not reuse symbol without anyone noticing.
Layout tips: keeping labels readable in small spaces
Start with hierarchy, because a label with perfect symbols can still fail if the user cannot find the expiry date. Put the most time sensitive and safety related items where the eye lands first, then group supporting details around them.
Keep symbol sizes consistent within a panel, and do not shrink icons until they look like noise. If you have to choose, drop low value symbols before you compromise legibility of the sterile symbol or the do not reuse symbol.
Whitespace is not wasted space, it is what makes scanning possible under bad lighting. A cramped cluster of ISO 15223-1 medical device symbols can look compliant on a monitor and become unreadable once printed on textured stock.
Use alignment rules that your team can repeat, like a fixed grid or consistent margins relative to the label edge. When artwork gets revised, those rules prevent the slow drift where the manufacturer symbol ends up on one panel and the address ends up somewhere else.
Decide early whether symbols will sit in a row, a column, or grouped by theme, and then stick to it. Consistency helps users build muscle memory across SKUs, which is the whole point of standardization.
Keep the relationship between a symbol and its data tight, especially for lot and date fields. If the symbol is far away from the printed value, people will mis-associate the number with the wrong meaning.
Watch contrast and background patterns, because subtle brand textures can destroy fine line icons. If you want a patterned label, reserve a clean white panel for ISO 15223-1 medical device symbols and key text.
Avoid rotating symbols to fit odd spaces, because users do not expect icons to be sideways. If you must rotate the layout, rotate the whole panel rather than individual elements.
Do not let barcodes bully everything else off the label. If the UDI carrier takes half the panel, consider moving it to a dedicated area so symbols and human readable text remain clear.
For curved surfaces like vials or small tubes, test readability on the actual diameter. A symbol that looks fine on a flat proof can become distorted once wrapped around a small radius.
Use clear separation between clinical symbols and logistics marks like “this way up.” If they blend together, users may treat everything as shipping content and ignore it in the clinical setting.
Pay attention to the edge of the label and sealing areas, because pouches and cartons have zones that get folded, heat sealed, or scuffed. A sterile symbol printed too close to a seal can become partially unreadable after packaging.
Finally, remember that readability is a system outcome, not a single design choice. The best layout is the one that survives printing variation, handling wear, and rushed users.
Managing symbol versions and keeping artwork consistent
Symbol errors often come from version sprawl, not from bad intent. Someone grabs a PNG from an old project folder, another person pulls an SVG from a vendor, and now you have two sterile symbol drawings that do not match.
Lock down a controlled symbol library with approved source files, naming conventions, and a single owner. If you treat ISO 15223-1 medical device symbols like controlled documents, you stop arguing about which file is “the right one.”
Consistency also means consistent stroke weight, corner radius, and clear space, especially when symbols sit next to each other. A manufacturer symbol with a different line weight than the rest makes the label look sloppy and invites questions during audits.
Track symbol changes the same way you track label copy changes, with revision history and a reason for change. When a complaint comes in, you want to know exactly when the do not reuse symbol was added or removed and who approved it.
Make the library usable, not just controlled, because people work around systems that slow them down. If the approved symbol folder is hard to access, someone will recreate the icon from memory in Illustrator.
Include guidance on minimum size and clear space in the library, not just the artwork. A perfect SVG still fails if it is printed at a size that turns fine details into blobs.
Decide whether you will allow filled icons, outline icons, or both, and document that rule. A mix of styles can happen slowly over time until the label looks like it was assembled from unrelated sources.
Keep an eye on file formats and conversions, because some workflows flatten vectors without warning. If your regulatory group approves an SVG but the printer receives a low resolution bitmap, you have a hidden process gap.
Build symbol checks into your artwork templates so the right icons are already present. Templates reduce the chance that someone forgets the manufacturer symbol when building a new SKU under time pressure.
When you update symbols, communicate the change to every team that touches labels, including suppliers. A controlled library does not help if contract packers keep using last year’s artwork package.
Audit your own labels periodically, because drift happens even with good controls. A quarterly spot check across product families can catch mismatched sterile symbols before an inspector does.
Also control how symbols are used in marketing collateral, because those files often leak back into packaging. If sales uses a nonstandard do not reuse icon in a brochure, someone will eventually paste it into a label layout.
Consistency is not about aesthetics, it is about reducing cognitive load for users and reviewers. When symbols look the same everywhere, people trust the label and move faster with fewer mistakes.
How to select symbols based on risk and user behavior
I have seen teams over label low risk accessories and under label high risk sterile products, and both choices confuse users. Use your risk management file to decide which user mistakes are realistic, then label to prevent those mistakes.
For example, if a device looks like it can be reprocessed, the do not reuse symbol earns its space. If a device is sterile and the sterile barrier is easy to compromise, storage and handling symbols become more important than marketing driven icons.
Think about who touches the package, because the first user is often a receiving clerk, not a clinician. If the outer shipper needs handling marks, keep them separate from ISO 15223-1 medical device symbols intended for clinical decisions.
Also consider training level, because a symbol that is obvious to a regulatory specialist can confuse a busy tech on a night shift. If your post market feedback shows repeat questions about sterility or reuse, treat that as a labeling signal.
Risk based selection works best when you connect each symbol to a hazard scenario. If the hazard is “use of nonsterile device in a sterile field,” the sterile symbol and barrier integrity cues become primary controls.
User behavior matters because people do not read labels like auditors do. They scan for a few anchors, like a sterile symbol, a date, and a lot number, and then they move on.
Look for “look-alike” risks where your product resembles a different one with different constraints. If two products share packaging and one is reusable, you need symbols that prevent mix-ups at the shelf.
Consider whether the device is used in emergencies, because stress changes attention. In urgent settings, clear symbols can outperform long text, but only if the symbol set is minimal and consistent.
Also consider the consequences of a wrong assumption, not just the probability of misuse. If reuse would create a serious infection risk, the do not reuse symbol should be prominent and hard to miss.
Storage symbols are often treated as secondary, but they can be the primary risk control for temperature sensitive materials. If performance degrades silently, the label has to prevent improper storage rather than trying to detect it later.
Think about the full lifecycle, including returns, rework, and field corrections. A clear manufacturer symbol and traceability fields help you manage post-market actions without guessing which unit is which.
If you have human factors data, use it to justify symbol choices and placement. Even informal feedback from clinicians can reveal which icons are ignored and which ones actually change behavior.
When in doubt, prioritize symbols that prevent irreversible actions. Once a device is opened, used, or reprocessed, you cannot undo the decision, so the label has to be strong before that moment.
Internal review steps to catch misuse before printing
Most symbol mistakes slip through because reviews focus on spelling and UDI formatting, not meaning. Build a review step where someone reads the label as a user and asks if each icon matches the product claims.
Do a quick cross check between the label, IFU, and technical file, because mismatches are common. If the IFU says single use and the label lacks the do not reuse symbol, treat it as a real defect, not a minor cleanup item.
Include packaging engineering in the review, because they see issues that regulatory may miss. If a symbol sits in a seal area or fold line, it can disappear in the final pack.
Have quality verify that label claims match the DMR and production routing. If the label says sterile but the work order is for a nonsterile configuration, you have a serious configuration control problem.
Run a “symbol meaning” review where the reviewer states what each icon means out loud. This simple step catches the classic error where a team uses the wrong sterile method symbol because the pictures look similar.
Check adjacency and pairing, because symbols rarely stand alone. A use by symbol without a clearly formatted date is not useful, and a lot symbol without a lot value is just noise.
Confirm that the manufacturer symbol and address match what you have registered with regulators. If the legal manufacturer name changes, labels often lag behind unless you force the check.
Make sure reviewers look at the label at actual size, not zoomed in on a screen. Zoom hides spacing problems and makes tiny icons look readable when they are not.
Do an ink spread reality check by printing on a similar printer and stock, even if it is not perfect. If thin lines fill in on a desktop proof, they will probably be worse on a high speed press.
Finally, confirm that the final approved PDF is the one that goes to production. Many labeling failures come from sending the wrong file version, not from approving the wrong design.
- Verify sterile symbol matches sterilization method and validation
- Confirm do not reuse symbol aligns with intended use and reprocessing guidance
- Check manufacturer symbol points to the legal manufacturer, not the packer
- Compare lot and use by format against ERP and UDI records
- Print a physical proof at final size and inspect under typical lighting
- Confirm symbol artwork matches the controlled library file names
If you can only add one more check, add the physical proof review at final size. It is the fastest way to catch the gap between “looks fine in design” and “fails in the real world.”
Document the review outcome in a way that is easy to retrieve later. When an auditor asks why a symbol is present or absent, you want a clear rationale rather than a vague memory.
Over time, track recurring findings and treat them as process issues. If every other label review catches a missing do not reuse symbol, your template and training need improvement.
Working with suppliers and printers on symbol fidelity
Your printer can ruin a clean symbol set with the wrong conversions, especially when they rasterize vectors or substitute fonts. Send vector artwork when possible and specify minimum line thickness so the sterile symbol does not fill in on press.
Ask for a contract proof that matches the real substrate, because glossy film and porous paper behave differently. A manufacturer symbol that looks fine on white paper can lose contrast on tinted Tyvek or a matte pouch.
Be explicit about color rules, since many regulatory labels are one color by design. If a supplier decides to “improve” contrast by changing symbol fill or reversing colors, you can end up with artwork that no longer matches your approved library.
When you outsource kitting, include symbol placement checks in the packaging specification, not just in email threads. The fastest way to get drift is to let each supplier build their own label template from scratch.
Give suppliers a single “artwork package” that includes the controlled symbols, the label layout, and the rules for use. If you send pieces in separate emails, you increase the chance they assemble the wrong combination.
Ask printers how they handle line art and whether they apply trapping or stroke expansion. Those normal print practices can change how ISO 15223-1 medical device symbols look at small sizes.
Confirm the printer’s minimum resolution and plate capabilities match your smallest symbol details. A symbol with fine gaps may close up if the press cannot hold the detail on your chosen substrate.
Include acceptance criteria for symbol legibility in incoming inspection when it makes sense. If you only inspect barcodes and text, symbol degradation can slip through until a user complains.
For thermal transfer or on-demand printing, validate the ribbon and printer settings. A do not reuse symbol printed with low heat or the wrong ribbon can look broken or incomplete.
Make sure suppliers understand which elements are regulated and cannot be moved casually. If they shift the manufacturer symbol to make room for a seam, they may unintentionally break your approved layout.
When you have multiple printers, harmonize their output with a shared proofing standard. If one site prints darker and another prints lighter, symbols can appear inconsistent across markets.
Finally, treat supplier feedback as useful data, because they see print failures early. If a printer says your minimum line thickness is too thin for a pouch film, adjust the symbol size or process before it becomes a deviation.
Keeping symbol use aligned with global regulatory expectations
ISO 15223-1 medical device symbols are widely recognized, but you still have to confirm what each target market expects on the label. Some regions care deeply about language requirements near symbols, while others focus on traceability fields and UDI placement.
Do not assume a symbol alone always satisfies an information requirement, because regulators may want accompanying text in certain cases. If your labeling strategy relies heavily on the sterile symbol and storage icons, document that rationale and keep it consistent across SKUs.
Watch for conflicts between standards, guidance, and local rules that influence packaging panels. If you add extra fields for one market, you may need to reflow symbols and then recheck minimum size and clear space.
When you create a global label, build a controlled “core panel” that rarely changes and a market specific panel that can change without breaking everything. That structure keeps the manufacturer symbol and key safety icons stable even as regulatory details evolve.
Build a market requirements matrix that explicitly lists which symbols are used and where. This keeps ISO 15223-1 medical device symbols from being added or removed based on assumptions during a fast launch.
Be careful with “global” labels that rely on a symbol legend in the IFU. If the IFU is not always present at the point of use, the legend strategy can fail even if it looks elegant on paper.
Some markets expect specific contact details, such as local representatives, and those details can compete with symbols for space. If you add addresses late, you can accidentally shrink symbols below a readable size.
Align your symbol approach with your language strategy so the label does not become a patchwork. If you use symbols to reduce translations, make sure the remaining text is consistent and minimal.
Keep an eye on how distributors apply overlabels for local compliance. Overlabeling can cover critical symbols or create a confusing double set of symbols that do not match.
Document your interpretation rules for ambiguous cases, like when a kit contains mixed sterility states. If you do not define the rule, each region or product team will solve it differently.
When regulations change, avoid reactive symbol churn that breaks label consistency. A controlled update plan helps you keep the core symbol set stable while still meeting new expectations.
Finally, train internal teams on the difference between “recognized” and “accepted without explanation.” Even when ISO 15223-1 medical device symbols are allowed, you still need to ensure users can interpret them correctly in your context.
Conclusion
ISO 15223-1 medical device symbols work best when you treat them like regulated content, not decorative graphics. If you control artwork, select icons based on real user decisions, and proof at final size, most labeling problems disappear before print.
Pay special attention to the sterile symbol, the do not reuse symbol, and the manufacturer symbol because those three drive high risk behavior and traceability. A clean, consistent label set builds trust with users and makes audits far less dramatic.
The practical win is that good symbol discipline reduces rework, deviations, and late stage artwork panic. When your process is stable, labels become a predictable output instead of a recurring fire drill.
If you want one takeaway, it is this: every symbol should earn its spot by preventing a real mistake. When you label with that mindset, ISO 15223-1 medical device symbols become a safety tool rather than a compliance checkbox.

